 
(b)Write the equation for the reaction between methanoic acid and aqueous sodium hydroxide. The structure below represents a portion of a polymer. (i) Write an equation for the reaction between propan-1-Ol and potassium metal. (c ) Study the scheme given below and answer the questions that follow. (i)What is the name given to the process? (b)Raw rubber is heated with sulphur in the manufacture of natural rubber (a)In which homologous series do the following compounds belong Describe how water could be used to separate a mixture of pentane and ethanol (b) In an experiment, excess ethanol was warmed with potassium dichromate for about twenty (ii) What is the name of the group of compounds to which Z belongs? (i) Name and draw the structural formula of compound Y. Y reacts with propanol to form a sweet smelling compound Z whose formula is. (a)When an organic compound Y is reacted with aqueous sodium carbonate, it produces carbon (IV) oxide. State the observation that would be made when a piece of sodium metal is placed in samples of: (b)The fermentation of glucose produces ethanol as shown in the equation below: State the type of reaction represented by: (i) and (ii). (a)The following equations represents two different types of reactions (b) Give the name of the organic compound formed when ethanol and propanoic acid react in the presence of concentrated sulphuric (VI) acid (iii)Determine the molecular formula of the hydrocarbon in (ii) above and draw its structural formula (ii) Predict the relative atomic mass of the hydrocarbon with 5 carbon atoms. (i)Write the general formula of the hydrocarbon in the table (b)Study the information in the table below and answer the questions Give the names of the following compounds: (ii) Choose two compounds which are members of the same homologous series and explain the difference in their melting points. 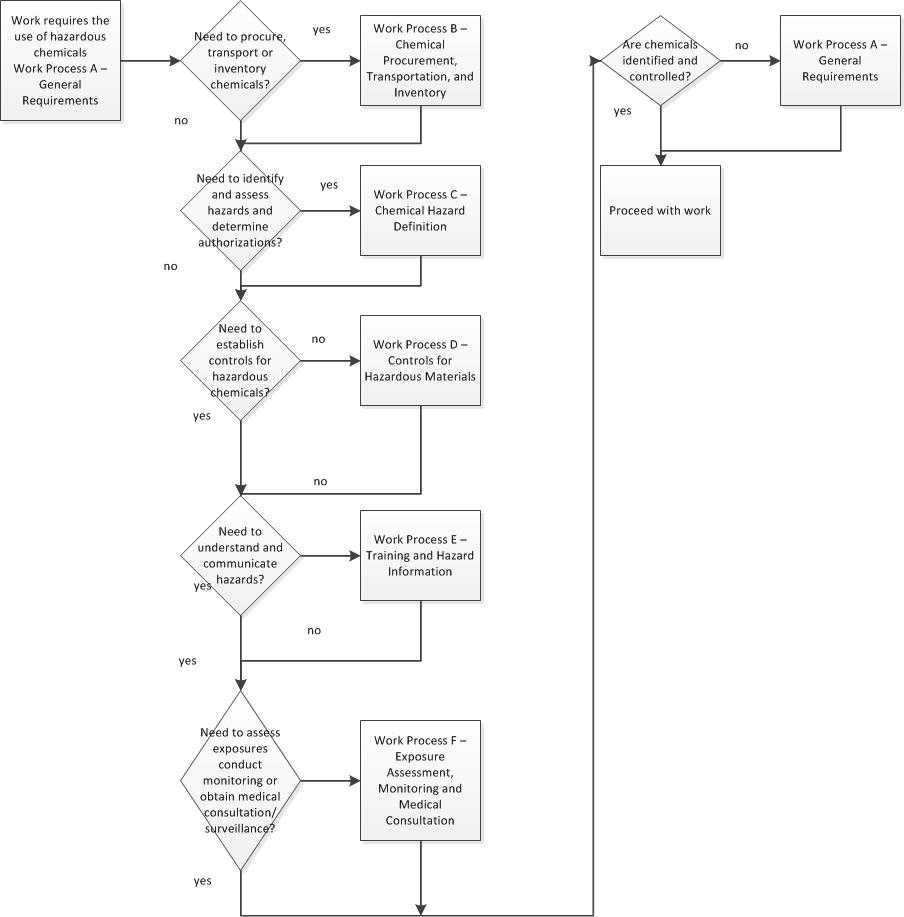
(i)Which of the compounds is a solid at #10.0^0#C? Explain. Study the table below and answer the questions that follow: This is because pentanoic acid reacts with sodium hydroxide leaving pentane which is neutral. 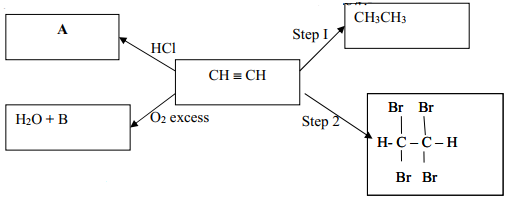
A mixture of pentane and pentanoic acid was shaken with 0.1 M sodium hydroxide solution and let to separate as shown in the diagram below: Determine the number of monomers on the polymer (H=1.0, C=12.0, N=14.0) A polymer has the following structure:Ī sample of this polymer if found to have a molecular mass of 5194. With reference to the structure, explain how the detergent removes grease during washing. A compound whose structure is shown below is found in a detergent. Explain how a sample of #CH_3CH_2CH_2OH#, could be distinguished from a sample of #CH_3COOH# by means of a chemical reaction. (b)Write an equation for the reaction between potassium and compound #C_4H_10O#. (a)Draw the structural formula and name the compound #C_4H_10O# A compound #C_4H_10O# is oxidized by excess acidified potassium manganate (VII) to form another compound #C_4H_8O_2#.The same compound #C_4H_10O# reacts with potassium to produce hydrogen gas. (b)Give one advantage of synthetic fibres over natural fibres. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
